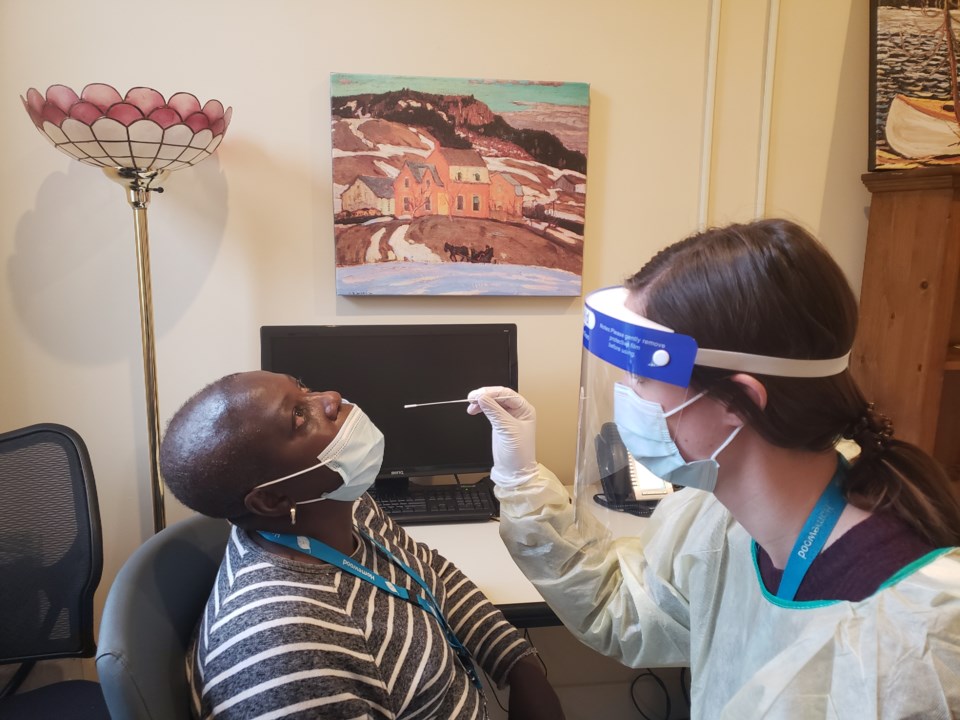
Hundreds of employees at Homewood Health Centre are part of a pilot project testing the effectiveness of rapid testing kits to detect COVID-19.
Homewood was recently selected by the Ministry of Health and Long-Term Care to be part of the pilot project to test Panbio COVID-19 Antigen Rapid Tests by Illinois-based Abbott.
The rapid tests take about 15 minutes to get a result and will be used as an enhanced screening method for some employees, said John Prieto, director of clinical support services, policy, and projects at Homewood.
Starting this week, the point-of-care tests have been used to screen about 500 of the more than 800 employees at Homewood, primarily within its front-line nursing and interdisciplinary professionals.
They will continue to be tested once per week.
“By doing this on a regular basis, we will be much better at identifying asymptomatic carriers of the virus and enhance our safety to our staff members and our patients,” said Prieto. “Staff are very excited they have this opportunity.”
Next week the program will expand to other staff members at the Guelph facility.
Homewood has over 300 beds in Guelph and offers mental health and addiction treatment services locally and throughout the country. Prieto said the facility is currently near capacity, despite the ongoing pandemic.
“We are sensitive and aware of the growing need, not just from COVID-19, but because of the many mental health factors that continue to impact our population and our community,” he said.
The pilot project will continue for eight weeks.
Prieto said the hope is by the end of the pilot all of the frontline staff at Homewood will have received the COVID-19 vaccine.
“We are currently in active conversations with Public Health to determine our vaccination prioritization list. That’s currently in works,” said Prieto.
The rapid tests are different than the PCR tests used by many COVID-19 testing clinics.
“The rapid antigen testing is not quite as accurate as PCR testing, but it is still considered highly accurate,” said Prieto. “I think that is partly why the ministry is interested in participating in this pilot, to help determine the utility of this the of testing in workplaces to enhance COVID-19 screening measures.”
The tests use a nasal swab to determine if someone is potentially infected with COVID-19 if a certain antibody associated with the SARS-CoV-2 is detected.
Positive results produce a visible line in the test area, not unlike a pregnancy test.
A positive result does not rule out bacterial infection or co-infection with other viruses and should be confirmed with additional testing and clinical correlation with patient history and other diagnostic information.
If someone at Homewood tests positive with the rapid tests they will be asked to perform a PCR test at the COVID-19 Assessment Clinic and the results of both tests will be shared with the ministry.
“We are committed to providing weekly updates to the ministry, how many tests are being conducted and how many positive or negative, how many are confirmed to be positive or negative through PCR testing,” said Prieto.